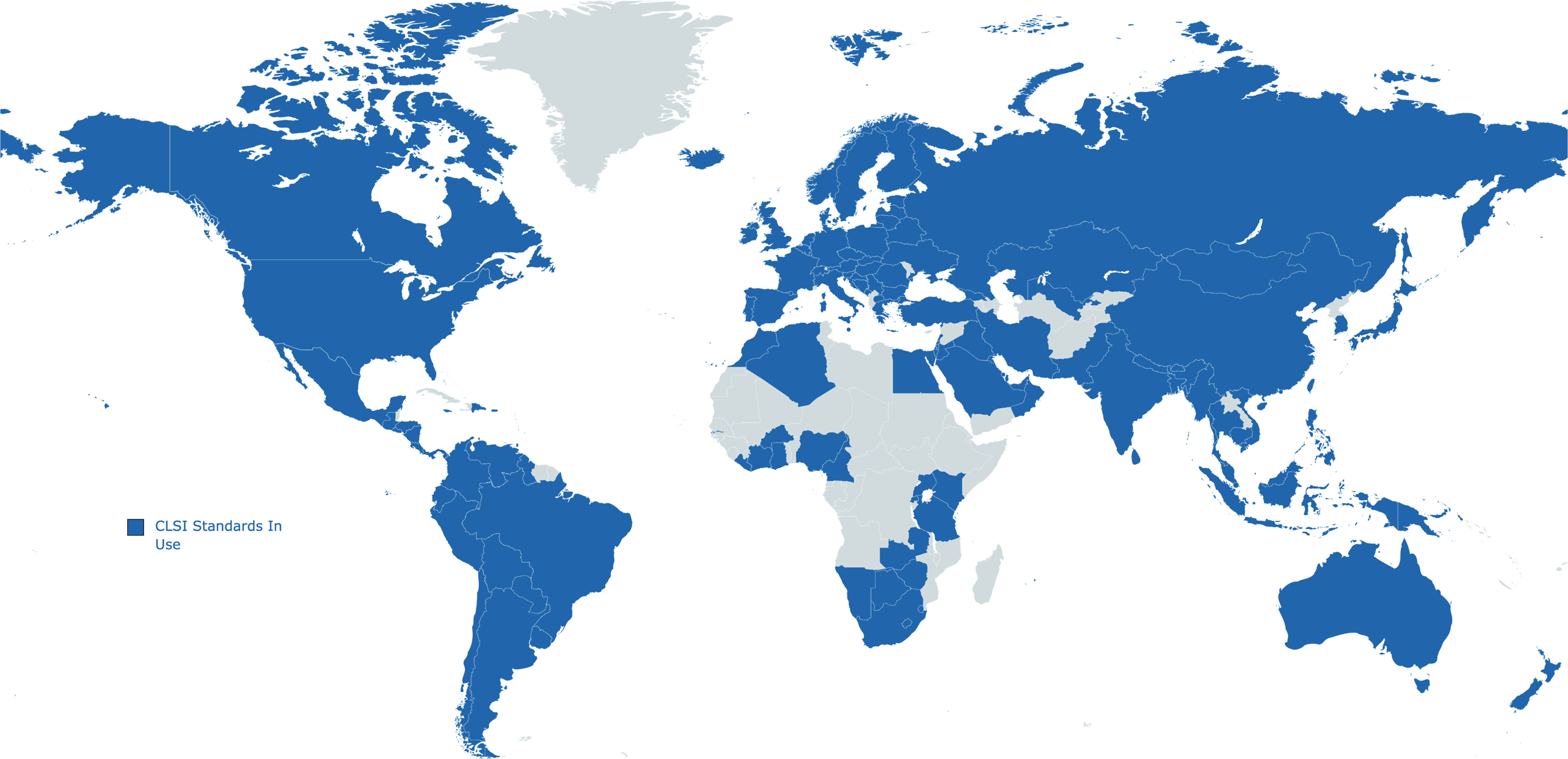
CLSI standards are recognized by laboratories, accreditors, and government agencies as the best way to improve medical laboratory testing. Standards go through rigorous development with contributions from industry, government, and health institutions. Discover how standards are developed.
More than 50 Years of Laboratory Leadership
Cultivating best practices and fostering innovation through standards and training

1967
Founding
Originally formed as the National Committee for Clinical Laboratory Standards (NCCLS), the organization was established to meet the emerging needs of the laboratory community and the technological revolution in laboratory products that grew in the 1960s. The rapid advancement in technology hastened the need for quality control and standardization in laboratory testing and created the basis for this collaboration between the US FDA, CDC, and College of American Pathologists (CAP), and many more under the leadership of Dr. Russell Eilers, the first CLSI president.

1969
First Standard Published
NCCLS published its first standard, Preparation of Manuals for Installation, Operation, and Repair of Laboratory Instruments, in 1969. During NCCLS’ first ten years, twenty laboratory standards were written.

1977
Accreditation
CLSI also became the home of the National Reference System for the Clinical Laboratory (NRSCL), a collection of standardized reference systems intended to make laboratory test results more uniform.

1985
Designated WHO Collaborating Center for clinical laboratory standards
By 1985, more than eighty standards had been written, more than 130 standards projects were underway, and more than 1,000 clinical laboratory scientists had participated in NCCLS standards projects. In the same year, as evidence of NCCLS’s international influence, the organization was designated as a World Health Organization Collaborating Center for Clinical Laboratory Standards.

1993
25th Anniversary
By its 25th anniversary in 1993, NCCLS had more than 1,800 active member organizations and institutions, and had published over 300 standards and guidelines.

1997
ISO Collaboration
In 1997 the International Organization for Standardization’s Technical Committee 212 (ISO/TC 212) was proposed and organized by NCCLS. Its purpose was to develop and harmonize voluntary international laboratory standards. Since then, CLSI has been responsible for managing ISO/TC212 and the US Technical Advisory Group (TAG) that facilitates US participation in the international standards development work.

2005
NCCLS becomes Clinical and Laboratory Standards Institute (CLSI) and launches Global Health Partnerships

2017
The FDA announces the recognition of numerous CLSI Standards.
Either in whole or part, which contain standardized reference methods and breakpoints that measure the susceptibility of bacteria and fungi to antimicrobial agents.

2014 - 2019
Global Development
CLSI focuses on global standards adoption and training in developing communities around the globe, assisting 51 laboratories in 8 countries to achieve and maintain international accreditation; conducting 327 mentorships in 20+ countries, and managing 102 QMS workshops that trained over 2000 laboratory professionals.

2020
CLSI launches MicroFree™
CLSI MicroFree™ Free platform allows easy access to critical antimicrobial susceptibility testing resources in service to the global efforts to combat antimicrobial resistance (AMR). The application wins recognition from ASAE for its global impact with the Power of Associations Gold Award in 2023.
2024
CLSI introduces Country-Based Pricing
Country-based pricing allows greater access to products and training for under-resourced countries through discounted pricing based on national income levels.

Today
CLSI is the leading global laboratory standards development organization with 292 standards and guidelines adopted in more than 155 countries around the world. Over 1,800 subject matter experts participate in standards development every year and our membership constituency is made up of more than 26,000 laboratory professionals.
Global Leader for Clinical and Laboratory Standards

Impact Around the Globe
CLSI standards are used by laboratories in 155 countries around the globe. Through our global partnerships program and philanthropic commitment, CLSI works to ensure laboratories around the world have the tools and training they need to succeed. By helping labs achieve sustainable quality, laboratories are able to better diagnose and treat patients and achieve better health outcomes.