CLSI’s evaluation protocol standards provide detailed explanations and instructions for the evaluation of test method performance characteristics such as precision and accuracy. These standards equip labs with the information needed to comply with the latest best practices and accreditation requirements.
In addition to evaluation protocol standards, CLSI has developed implementation and establishment guides and tools designed for use by laboratory scientists when performing verification and validation testing. Minimum protocols are provided in the establishment and implementation guides, including how to select appropriate samples and analyze data.
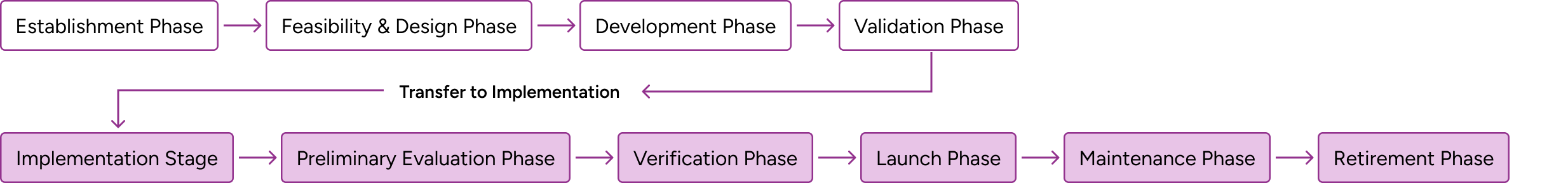
CLSI method evaluation guidance supports developers of both commercially available in vitro devices (IVDs) and laboratory developed tests (LDTs), and follows the life of a test through establishment and implementation. CLSI EP19 presents a framework that all CLSI evaluation protocol standards fall into.
CLSI guidance is developed through a robust consensus process that includes the open and transparent input of all affected stakeholders from industry, government, and professions.