You'll receive the latest updates on new standards, guidelines, and educational resources, as well as expert insights to help enhance your laboratory's performance and compliance.
September is Newborn Screening Awareness Month
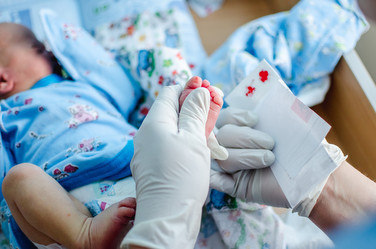
September is Newborn Screening Awareness Month. 2020’s theme is “Expect newborn screening.” Newborn screening is a state public health service that reaches each of the nearly 4 million babies born in the United States each year. It ensures that all babies are screened for certain serious conditions at birth and for those babies with those conditions, it allows doctors to start treatment before some of the harmful effects happen.
Although newborn screening requirements vary from state to state, most states test for all the conditions specified by the Health Resources and Services Administration (HRSA) in their Recommended Uniform Screening Panel. These conditions include phenylketonuria (PKU), cystic fibrosis, sickle cell disease, critical congenital heart disease, hearing loss, and others. Some states test for additional disorders that are not part of the HRSA panel.
Most of the conditions included in newborn screening can cause serious health problems if treatment is not started shortly after birth. Prompt identification and management of these conditions may be able to prevent life-threatening complications.
Newborn blood spot screening, the method by which inherited and metabolic disorders are tested in newborns, involves a small prick on the baby's heel and a few drops of blood placed onto a screening card. Testing is usually performed anywhere from 24 to 48 hours of age, with some states requiring follow-up by a pediatrician or other health care provider several weeks after birth. Standardization in methods such as newborn dried blood spot screening improves testing accuracy, resulting in better patient outcomes.
CLSI’s newborn screening standards provide test collection instructions, as well as information on methods for the detection of metabolic and hereditary disorders. Ensure your lab has the information needed to perform safe and timely newborn screening. For more information visit https://clsi.org/standards/products/newborn-screening/.